Several diagnostic methods are available for grapevine virus identification. In this second article in our series to improve grower knowledge of viruses, Dr Nuredin Habili from Waite Diagnostics, discusses these methods, their benefits and their limitations.
The methods available include:
- Field observation of symptoms;
- Long term biological indexing;
- Short term molecular indexing using ELISA;
- Short term molecular indexing using PCR; and
- Short term molecular indexing using High Throughput Sequencing
Field observation of symptoms
Before conducting any laboratory testing, ‘symptomatology’ (searching for identifiable symptoms) in the field is the first step in diagnosing the cause of any disorder.

Generally, we find that virus symptom expression is most obvious in late autumn, however this timing can vary based on the virus in question. This technique involves you knowing what to look out for, or at least recognising potential virus-like symptoms as opposed to nutritional or other disorders. Articles defining visual symptoms for a range of viruses will follow.
Most grapevine virus diseases, especially leafroll, produce symptoms on sensitive red varieties. Leafroll is a virus disease which has obvious symptoms of red leaves with green or yellow veins with downward rolling of older leaf blades (Figure 1). Detection of the ‘type’ of leafroll virus associated with the reddening is only possible by molecular analysis.
A recent development relating to the use of symptomatology for large scale detection of GLRaV-3 (LR3) in Napa Valley (California) is recording the rate of the LR3 infection by aeroplane using hyperspectral imaging technology (MacDonald et al, 2016).
In autumn 2014, more than 21,500 vines from five vineyards of Cabernet Sauvignon were analysed and showed an infection rate between 6 to 80%. It was demonstrated that the method had a success rate of 94% in virus detection, and it had only 4.7% disagreement with the lab testing of the virus.
If this technique is undertaken for a few consecutive years, the rate of infection and the direction of spread may be determined. Trials are also underway in Australia looking to detect viruses using imaging.
Long-term biological indexing
This is another application of ‘symptomatology’ where specific woody indicators are widely used. This technique involves grafting a bud from a plant suspected to carry a virus onto a series of sensitive woody indicator plants. Each woody indicator reacts differently to a virus pathogen. After 1 to 3 years, the inoculated plants may show virus disease symptoms.
This indexing technique can sometimes also be performed using herbaceous host plants which need less time than woody plants to give results. For example, Grapevine fanleaf virus can be detected by inoculating the sap from a suspected host plant to a herbaceous indicator, e.g. Chenopodium, and waiting for the symptoms to appear within two weeks.
Short term molecular indexing
ELISA

This approach of indexing harnesses the biochemical nature of a virus particle which consists of just a few functional genes (one or more pieces of RNA or DNA) wrapped within the coat protein. Antibodies produced against a specific virus can be used in an immunological assay platform, the most famous of which is called ELISA (enzyme-linked immunosorbent assay). ELISA incorporates separation of specific and non-specific interactions via serial binding to a solid surface, usually a polystyrene multi-well plate. This results in a coloured end product which correlates to the amount of analyte present in the original sample (5) (Figure 2).
ELISAs have the benefit of being quick and simple to carry out, and are designed to rapidly handle many samples in parallel. Sensitivity can, however, be improved using alternative methods of molecular indexing.
PCR and HTS
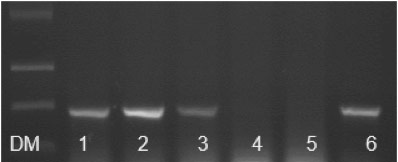
The detection methods that employ viral nucleic acids (RNA or DNA) can be up to 100 times more sensitive than ELISA. The nucleic acid-based methods are available in different formats and are being evolved by becoming more sensitive, rapid and specific. Of these, Reverse Transcription – Polymerase Chain Reaction (RT-PCR) using either end-point (Figure 3), multiplex or real time PCR are more common. These methods are designed to detect the virus itself, rather than the presence of antibodies (generally), as for ELISA. Microarrays and Lamp methods are gaining popularity, while ‘High Throughput Sequencing’ (HTS), also known as ‘Next Generation Sequencing’, is the ultimate method of choice which can rapidly detect the virome (the collection of viruses) in a vineyard (Saldarelli et al., 2017).
HTS generally comes into consideration when neither the ELISA nor PCR testing has returned a positive for a known virus, when the plant is symptomatic. Since its discovery, the HTS technique has added a few emerging viruses to the list of 65 already detected in the grapevine; including Grapevine Syrah virus-1, Grapevine red blotch virus, Grapevine Pinot Gris Virus and Grapevine enamovirus-1, a Luteovirus from Brazil (Silva et al., 2017). Some of these viruses like Grapevine virus E and Syrah virus-1 have not been associated with any disease, and apparently do not impose any biosecurity risk.
There are a few laboratories in Australia with the capability of undertaking HTS, however testing is currently expensive due to the required infrastructure such as computer capacity and storage and personnel expertise to comprehensively analyse and interpret the data.
A drawback associated with HTS is the detection of a number of sequences related to viruses which are likely to have been integrated in the plant chromosome for thousands of years (Geering et al., 2014). These can be wrongly considered as pathogenic to grapevines. It is recommended that any virus in grapevines detected by HTS be graft transmissible, and its full sequence be determined prior to claiming pathogenicity.
References
- Geering et al., (2014). Endogenous florendoviruses are major components of plant genomes and hallmarks of virus evolution. Nature Communications. 5:5269 | DOI: 10.1038/ncomms6269.
- MacDonald, S. L., Staid, M., Staid, M. and Cooper, M. L. (2016) Remote hyperspectral imaging of grapevine leafroll-associated virus 3 in cabernet sauvignon vineyards. Computers and Electronics in Agriculture, 130, pp. 109-117.
- Saldarelli, P., Giampetruzzi, A. Maree, H.J. and Al Rwahnih, M. (2017). High-Throughput Sequencing: Advantages beyond virus identification In: Grapevine Viruses: Molecular Biology, Diagnostics and Management, Meng, B. et al. (eds.), 625-642.
- Silva et al. (2017). Discovery and Molecular characterization of a novel enamovirus, Grapevine enamovirus-1. Virus genes 53: 667-671.
- Introduction to ELISA – Basics Guide. https://www.bio-rad-antibodies.com/an-introduction-to-elisa.html
Dr Nuredin Habili is an experienced plant virologist with more than 20 years’ experience working with plant viruses. Through Waite Diagnostics he offers virus testing to customers all over Australia, as well as the United States of America, New Zealand, Germany, Argentina and South Africa. For further information, please contact Dr Nuredin Habili, Waite Diagnostics, School of Agriculture, Food & Wine, The University of Adelaide at Nuredin.habili@adelaide.edu.au